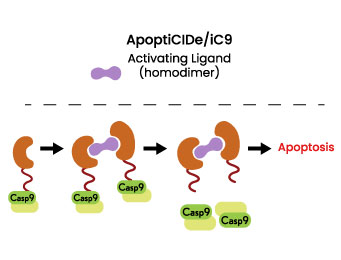
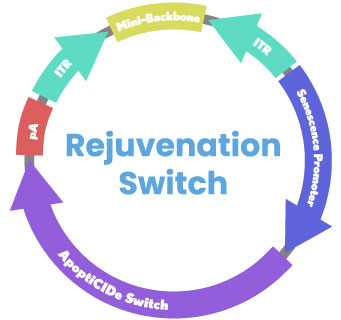
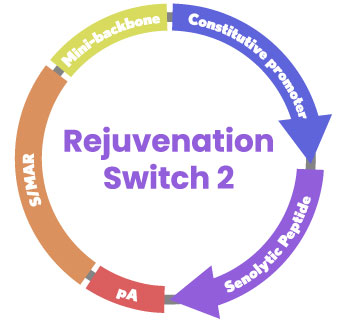
Mechanism:
There are many options for transdermal DNA delivery, including intradermal injection, electroporation, Gene Gun biolistic particle delivery, and microneedles (MNs), which come in several different forms, such as microchannels, coated, and dissolving MNs, (comprising biodegradable polymers). We plan to use dissolving MNs, due to higher loading payload capacity and ease of use. Although we plan to apply it over a relatively small area, others have shown the practicality and safety of application to a much larger area, including the face in cosmetic applications (before/after shown).
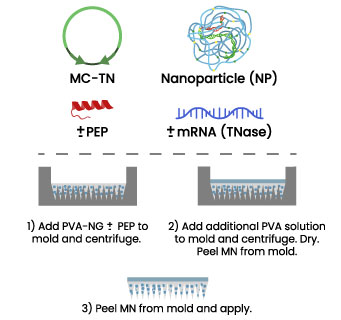
As a lead option, we will load NPs with mini-plasmid along with a tissue permeability-enhancing peptide (PEP) ± transposase mRNA and embed it in a biodegradable Polyvinyl alcohol (PVA) MN mold (shown).